Teva's ProAir Digihaler FDA Approved to Monitor Asthma and COPD Treatment - Basel St 5, Petah Tikva, Israel

Teva Pharmaceuticals Announces Launch of Daily Maintenance Asthma Medication | Asthma and Allergy Foundation of America

Teva's generic of AirDuo RespiClick® Inhalation Powder: Fluticasone Propionate and Salmeterol Inhalation Powder (Multidose Dry Powder Inhaler)

Teva Launches Two Digital Inhalers in the U.S., AirDuo® Digihaler® (fluticasone propionate and salmeterol) Inhalation Powder and ArmonAir® Digihaler® (fluticasone propionate) Inhalation Powder | Business Wire

First and Only Digital Inhaler With Built-In Sensors Approved by FDA - Pharmaceutical Processing World

Teva Announces FDA Approval of First and Only Digital Inhaler with Built-In Sensors – ProAir® Digihaler™ | SnackSafely.com
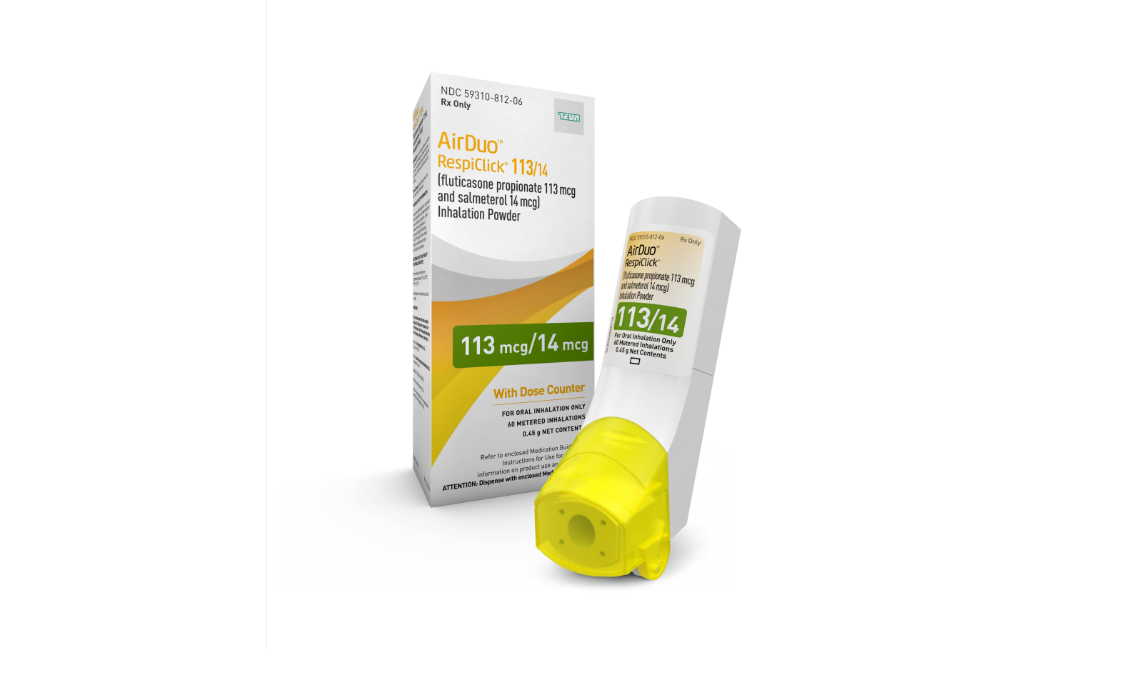
Teva Introduces Inhalation Powder and Generic Version in U.S. for Asthma Treatment - Drug Discovery and Development