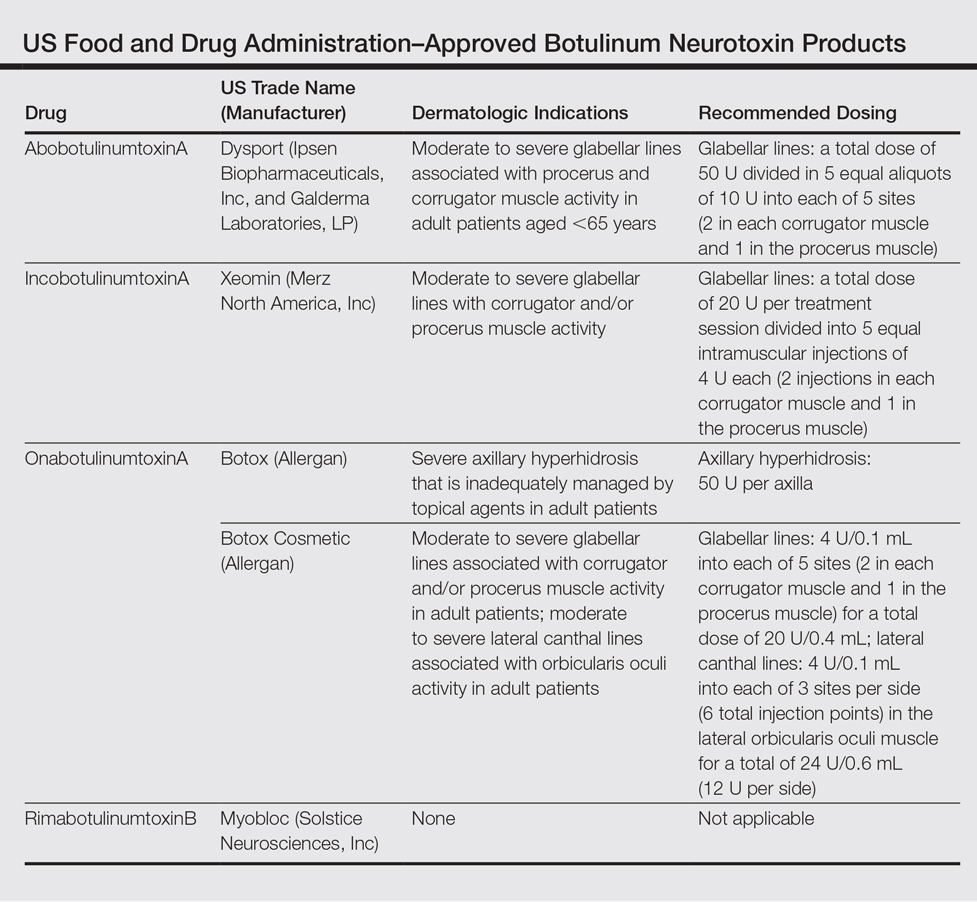
IPSEN: Announces FDA Approval of Dysport® (abobotulinumtoxinA) for the Treatment of Upper Limb Spasticity in Children, Excluding Cerebral Palsy | FDA Reporter

AbbVie : FDA Approves Expanded BOTOX Label for the Treatment of Pediatric Patients with Spasticity | World Pharma Today

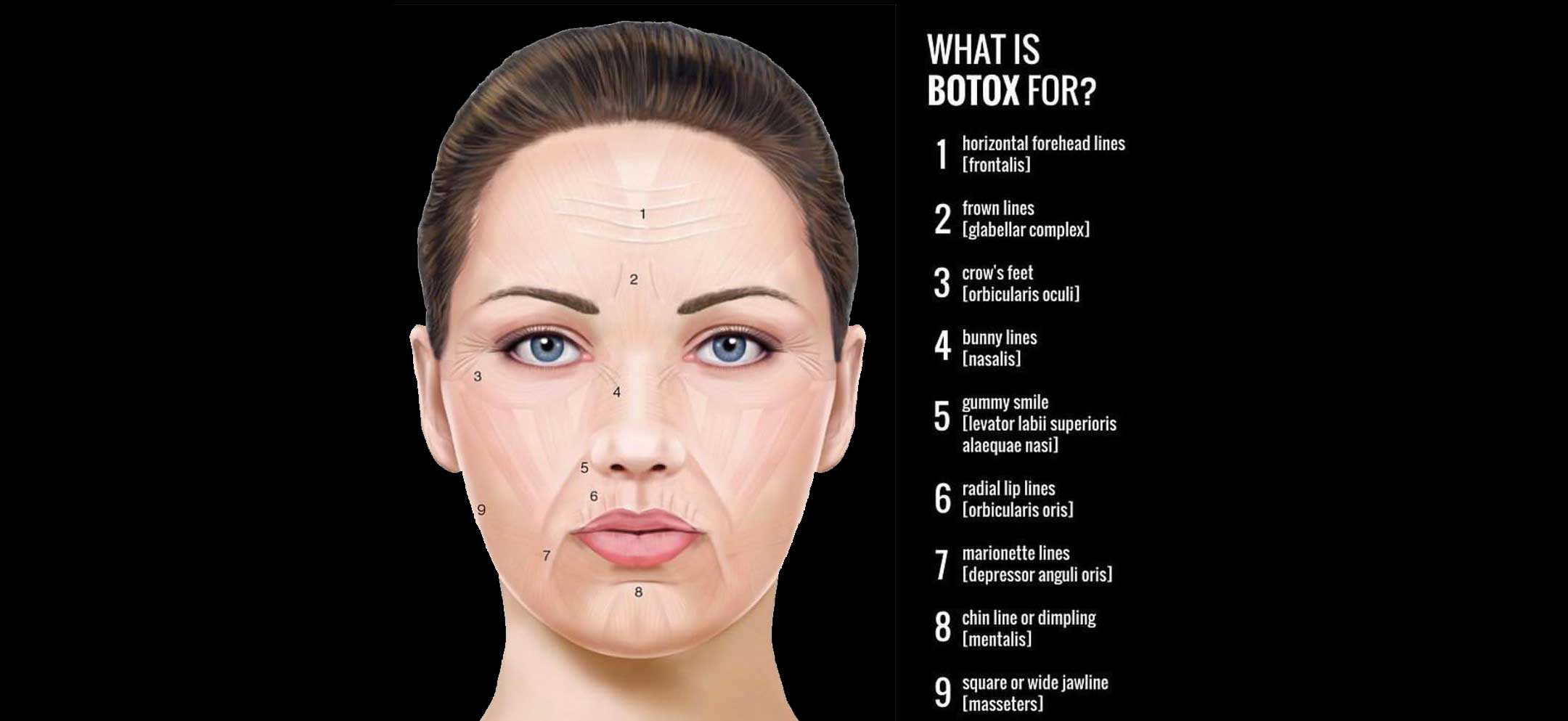
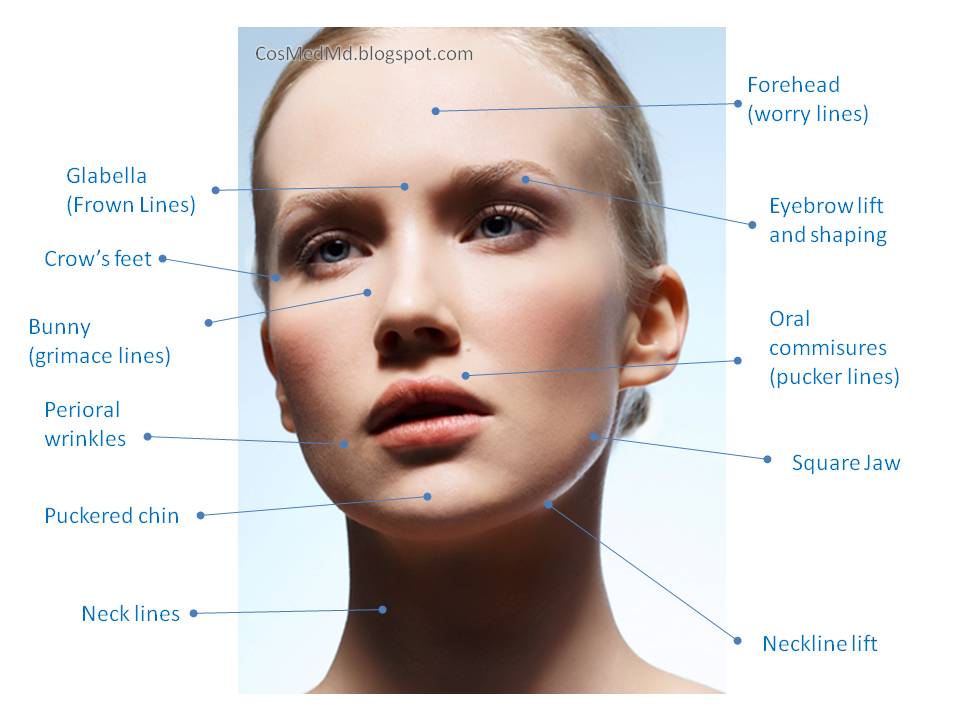
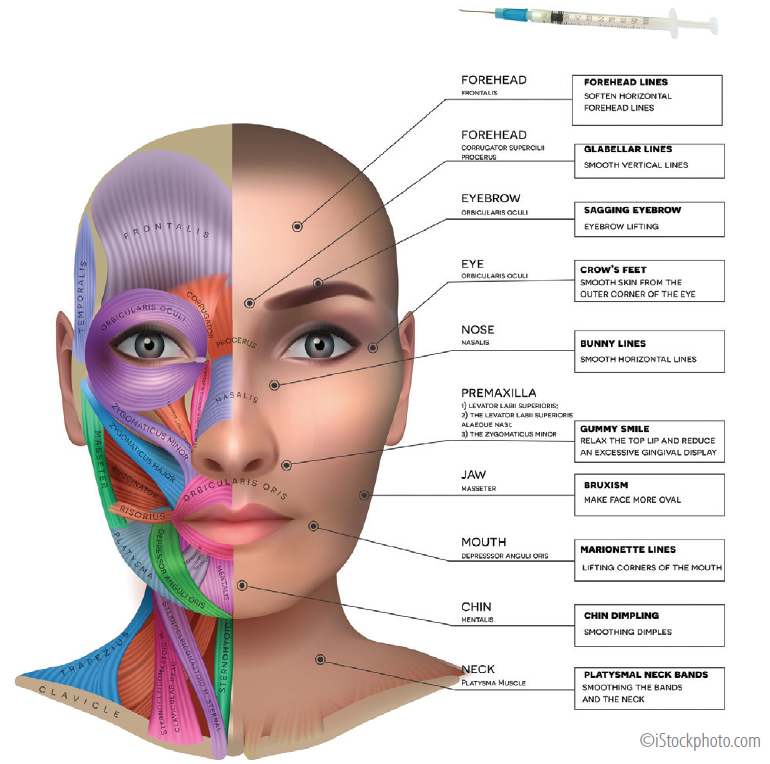
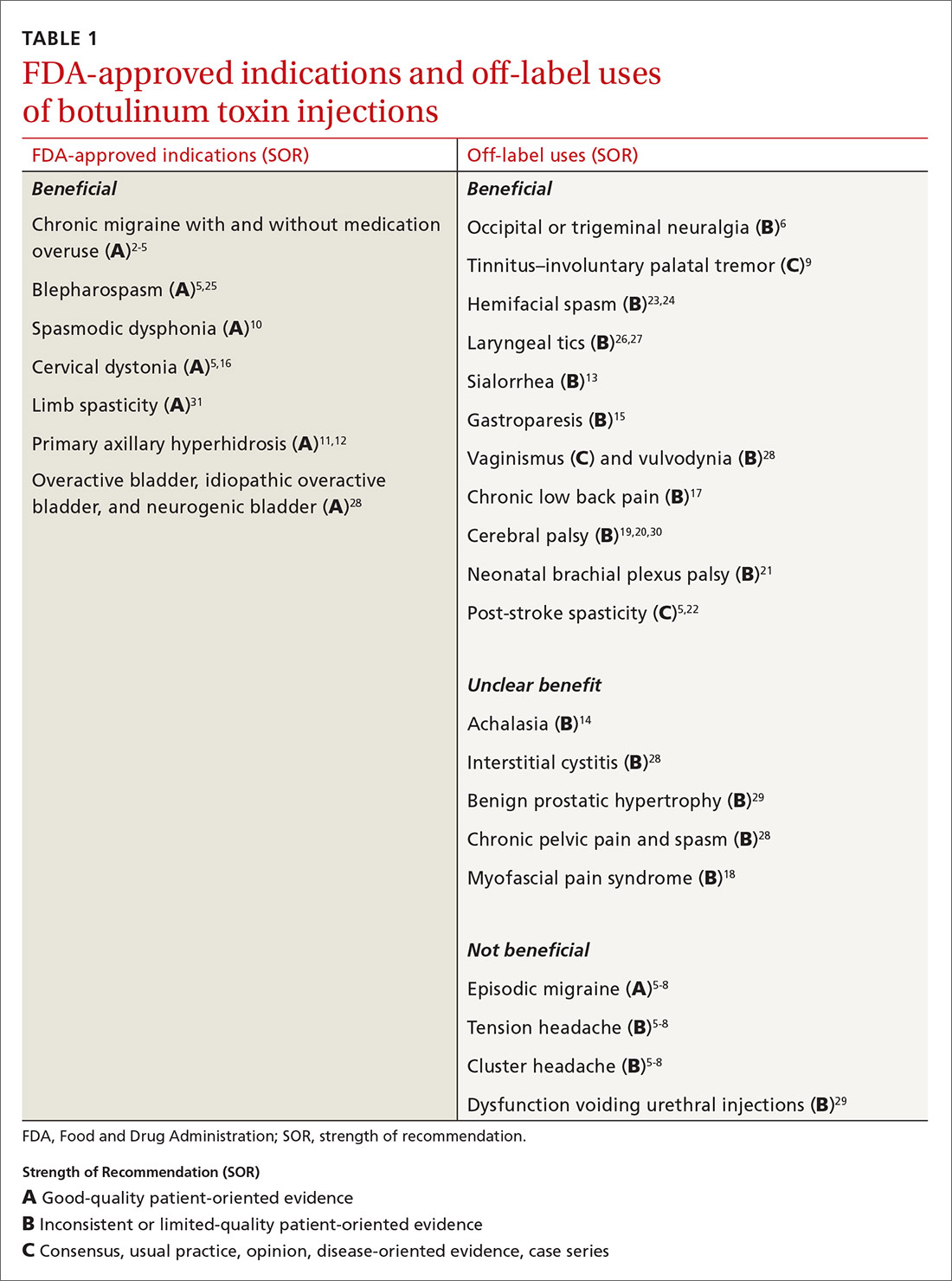
FDA Approves Expanded BOTOX® Label to Include Eight New Muscles to Treat Adults with Upper Limb Spasticity | News | Spinal Cord Injury Zone!